
US-based foot and ankle surgery devices maker Paragon 28 has launched a new allograft, the Preserve HammerGraft System for hammertoe and new charcot and midfoot reconstruction and the Gorilla Medial Column Plating System for medial column deformities.
The new hammergraft system expands the firm's existing line of Preserve implants that are aseptically processed without gamma radiation or bleaching.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
The system includes four allograft implants in 2.3mm and 2.8mm straight and angled options, which are sourced from the cortical rim of the tibia or femur to provide similar density to PIP joint, as well as strength and blood flow requirements of the region.
The diametre of the drill and ribs on the graft were sized to reduce crushing of cancellous bone and optimised to prevent pullout of the implant after insertion.
Packaged in a hydration solution, the cross section of the implant offers resistance to rotation, while the tapered ends facilitate easy insertion.
The system also comes with dense cancellous spacers in 6mm, 8mm and 10mm options to allow lengthening at the PIP joint.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
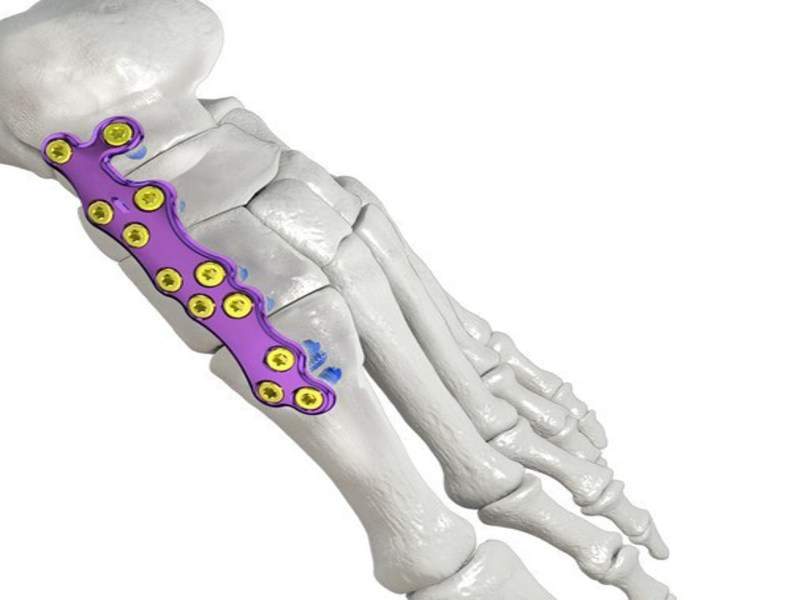
By GlobalDataThe Gorilla Medial Column Plating System includes 36 plating options in thickness options of 1.5mm and 2.0mm to accommodate different patient anatomies, levels of deformity as well as size and severity of condition.
The plates are made of titanium (Ti6AI4V-ELI) to enhance strength and accept 2.7, 3.5 and 4.2mm locking and non-locking polyaxial screws to allows access to highest quality bone around the fracture site.
They also accept both R3CON and TuffNeck screw technologies for flexibility of varying stoutness, and the holes have a built-in recess to minimise screw head prominence.
Image: Gorilla Medial Column arch plate. Photo: courtesy of Paragon 28.





