Visit our Covid-19 microsite for the latest coronavirus news, analysis and updates
Follow the latest updates of the outbreak on our timeline.

Discover B2B Marketing That Performs
Combine business intelligence and editorial excellence to reach engaged professionals across 36 leading media platforms.
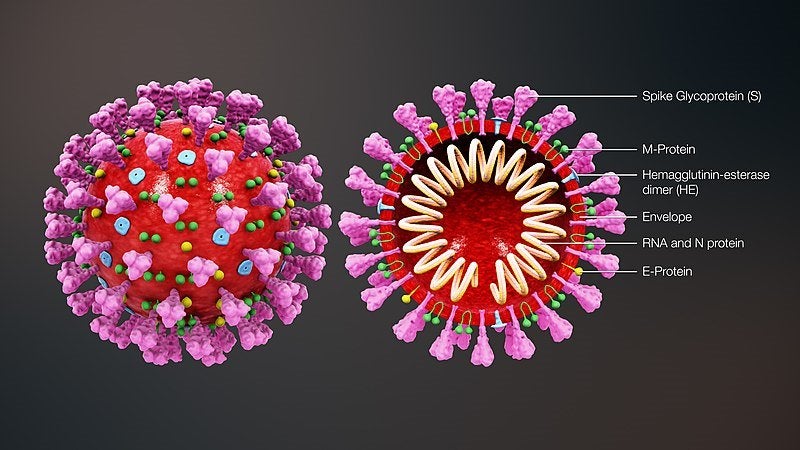
US-based Co-Diagnostics has launched a research use only (RUO) CoPrimer test for new coronavirus (2019-nCoV).
The company launched the product just weeks after it completed principle design work for the test to address the growing need for testing designs to detect the presence of coronavirus.
To date, the coronavirus outbreak killed 638 people, with a total of 31,428 infection cases now confirmed.
The Logix Smart 2019-nCoV coronavirus test is now available for sale to laboratories, hospitals and institutions.

US Tariffs are shifting - will you react or anticipate?
Don’t let policy changes catch you off guard. Stay proactive with real-time data and expert analysis.
By GlobalDataDesigned leveraging the company’s CoDx Design software system, the test helps in identifying the most optimal target on the 2019-nCoV genome for a PCR assay.
Co-Diagnostics expects that the test will ensure greater accuracy in detecting the presence of the coronavirus.
Co-Diagnostics CEO Dwight Egan said: “Leveraging our proprietary design process and software has allowed us to quickly move this product from design into commercialisation, and to do so with the confidence that our high-quality product meets our goal of providing an effective, much-needed global diagnostic solution in an emergency situation.”
“As a result of our rapid development, we have already received requests from customers in countries across the world to purchase tests.
“Sales and shipments of products will be fulfilled from our Utah headquarters to customers who have the capacity to utilise RUO products to slow the spread of this epidemic.”
Last week, Novacyt’s molecular diagnostics division Primerdesign launched a molecular RUO test for 2019-nCoV.
Meanwhile, the coronavirus outbreak, which originated from China’s Hubei province, has affected a total of 26 countries.





