Clarius Mobile Health and ThinkSono have introduced a new AI-guided ultrasound system designed to enhance the detection of deep vein thrombosis (DVT) in Europe.
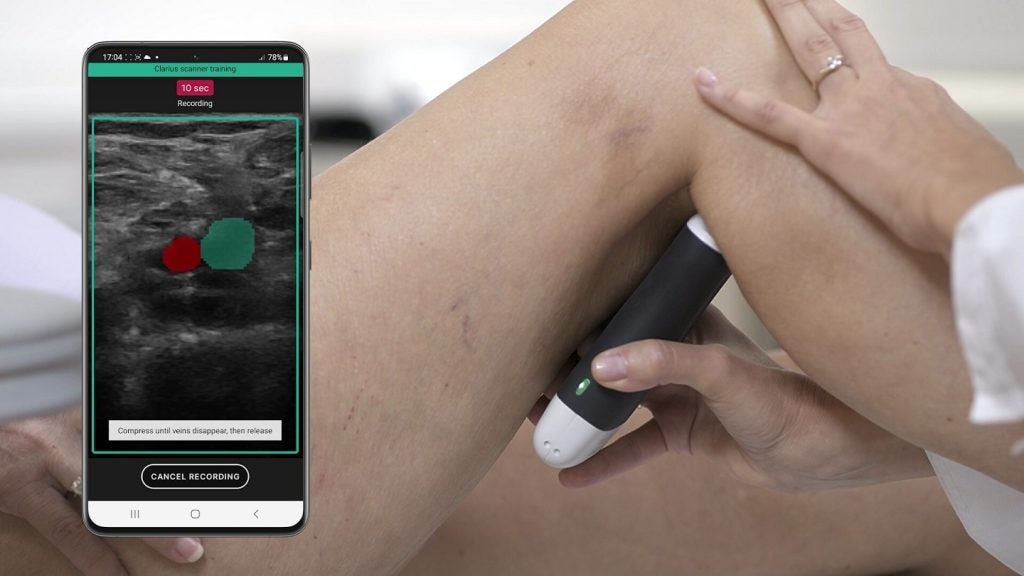
ThinkSono Guidance AI App, which pairs with the ultrasound scanners of Clarius, has received Class llb CE mark regulatory approval.
This app allows healthcare professionals to collect DVT exam data using the Clarius handheld ultrasound scanner.
A qualified clinician can then review the collected data, potentially reducing diagnosis time to as little as 15 minutes.
Further clinical studies are being conducted in the US at NYU Langone Health and Temple Health, following the successful European multi-site clinical trials that led to the CE mark approval for the ThinkSono Guidance System.
These US studies are part of the preparation for the Food and Drug Administration (FDA) submission.
ThinkSono's collaboration with Clarius began in 2017 when it purchased the first Clarius scanner and utilised the Clarius Cast API, aimed at researchers and AI companies. This collaboration has since evolved with ThinkSono now using the Clarius Solum SDK to develop a customised solution for its DVT application.
The complete DVT solution, including a Clarius ultrasound scanner, will be marketed and sold by ThinkSono in the UK, Germany, and Greece.
ThinkSono CEO and co-founder Fouad-Al Noor said: “Clarius has been an invaluable partner in helping us bring ThinkSono Guidance to market quickly to help non-ultrasound trained staff such as nurses improve the DVT pathway by scanning for DVTs at the bedside.
“Our app integrates seamlessly with Clarius scanners through the research SDK, which sends streams of real-time data to the ThinkSono App that, in turn, provides instant guidance to users as they perform an ultrasound exam. Our partnership with Clarius has been vital in running our clinical studies.”
The Clarius SDK suite is designed to facilitate the integration of AI-powered ultrasound software with Clarius handheld ultrasound systems, the Clarius Cloud exam management platform and the Clarius App.
Earlier this year, Clarius Mobile Health secured FDA 510(k) clearance for its Clarius Bladder AI non-invasive tool to measure bladder volume automatically.