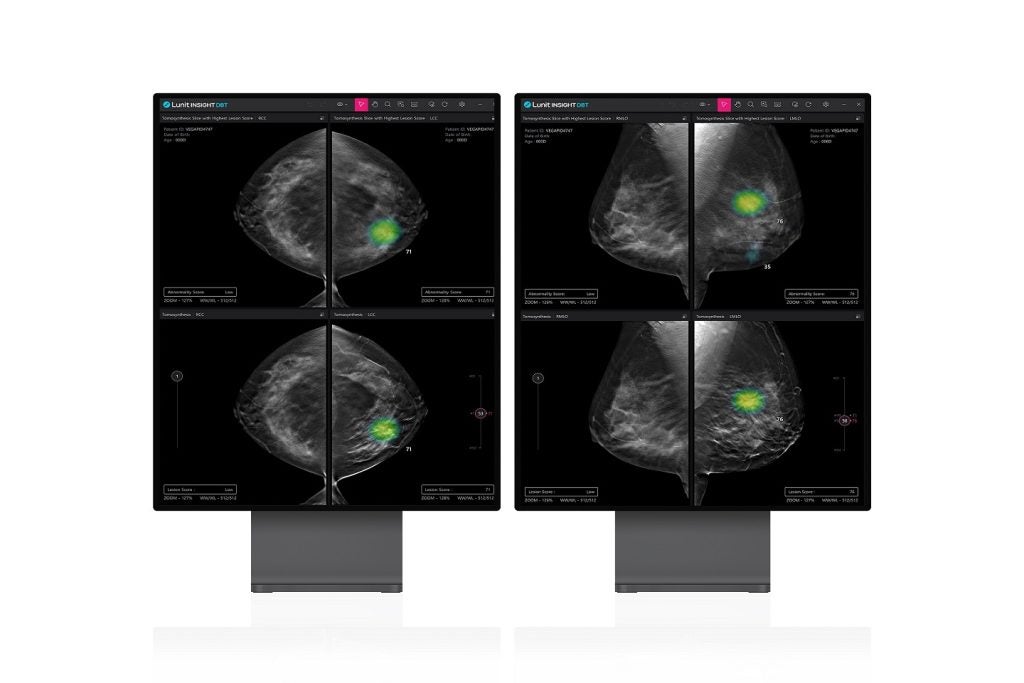
Lunit has received 510(k) clearance from the US Food and Drug Administration (FDA) for its 3D Breast Tomosynthesis (DBT) artificial intelligence (AI)-powered solution.
Dubbed Lunit INSIGHT DBT, the new AI algorithm is intended for analysing 3D images generated by DBT equipment.
Compared to traditional 2D mammography screenings, DBT imaging is said to provide a precise breast cancer diagnosis.
DBT's ability to offer accurate breast cancer screenings has led to a demand for this technology, particularly from advanced US medical institutions, accounting for more than 64% of the demand globally for DBT.
In March 2023, Lunit INSIGHT DBT received a CE mark under Europe's latest Medical Device Regulation (EU MDR).
Lunit CEO Brandon Suh said: “The US is the biggest player in the global breast screening market, accounting for up to 40% of the market share. More than 40 million mammography screenings are reported in the US annually.
“Given this substantial market influence, achieving FDA clearance for Lunit INSIGHT DBT not only solidifies our presence in the largest market but also marks a significant milestone in our mission to revolutionise breast cancer diagnosis and, ultimately, save more lives.”
Lunit, a medical AI company, is focused on the development of AI solutions for precision diagnostics and therapeutics.
In November 2021, the company also obtained clearance from the FDA for its Lunit INSIGHT CXR Triagechest X-ray triage solution and Lunit INSIGHT MMG AI-powered mammography analysis solution.