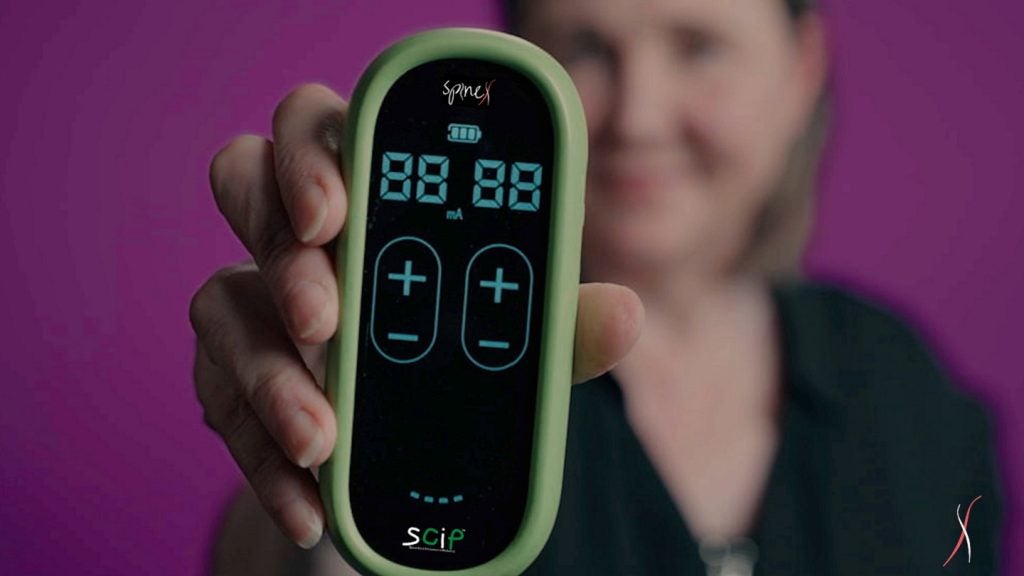
SpineX has received funds to complete a pivotal clinical trial of the SCiP device in children with cerebral palsy (CP), a movement disorder in childhood.
The National Institutes of Health, through the Small Business Innovation Research Awards, has provided the funding.
In the pivotal trial, the company will test the safety and efficacy of the SCiP device.
The data obtained from the trial will be used by the company to seek approval from the US Food and Drug Administration (FDA) for marketing and further development of the commercial device.
SCiP therapy targets the abnormal connectivity between the brain and spinal cord to treat the root causes of CP.
To date, the therapy has shown durable functional improvements that are significantly greater compared to the existing treatments in children with CP.
SCiP technology demonstrated its ability to treat the root cause of CP through spinal cord neuromodulation in investigational studies.
The company plans to initiate patient recruitment for the trial in the first half of next year and launch SCiP therapy in the US in 2026.
SpineX product director Kara Allanach said: “Not only is it a nod to our commercial viability, it provides us with the funding to complete our trial and move quickly to market.”
SCiP has previously received the breakthrough device designation based on pilot clinical results, which enabled the company to accelerate its pathway for regulatory clearance.